|
Recently, Abbott reported data from a new study showing that two blood-based biomarkers measured using its tests were able to predict a patient’s recovery from traumatic brain injury (TBI). The Proclaim Plus system also allows physicians to control multiple independent BurstDR stimulation areas, providing wide pain coverage while eliminating the risk of overstimulation. 4, 2022 / PRNewswire / - Abbott (NYSE: ABT) announced today that the U.S. It also noted that the FlexBurst360 therapy will allow physicians to identify the lowest effective dose of stimulation for each patient and adjust the stimulation depending on evolving pain needs. In the end, it provides a trial summary report for documentation. 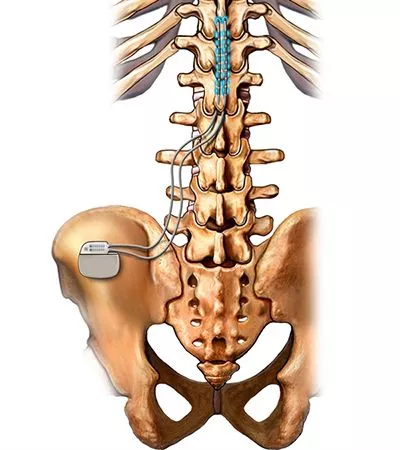
It starts with patient education and personalized goal setting, followed by simple progress tracking and real-time expert support. It also improved patients’ life quality and reduced the emotional suffering related to pain.Ībbott stated that the BurstDR technology offers pain relief in a sub-sensory range, which is not felt by the patient. Boston Scientific can help you maximize your patients’ chances for successful therapy with mySCS. In clinical trials, the technology provided superior pain relief compared to tonic stimulation. 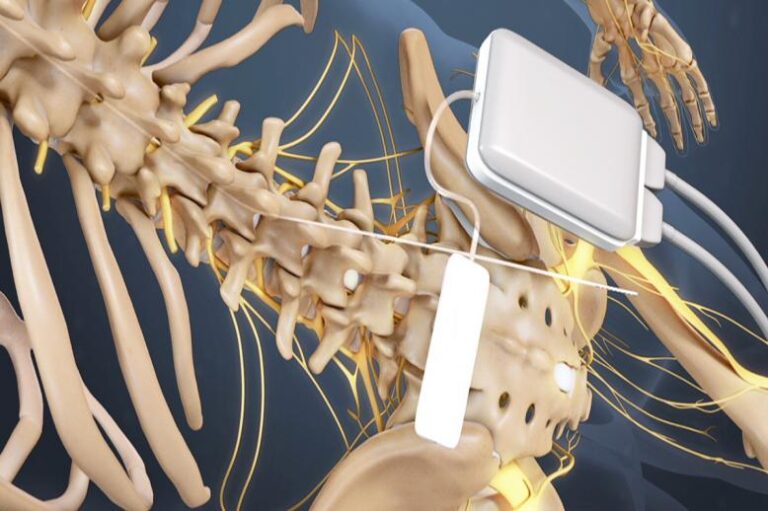
The company’s exclusive stimulation technology, BurstDR therapy delivers pulses, or bursts, of mild electrical energy to change pain signals as they travel from the spinal cord to the brain. Initially, spinal cord stimulators were implanted by spine surgeons, necessitating general anesthesia, laminectomy, electrode placement directly onto dura, and postoperative hospital admission. “Our latest development, Proclaim Plus with FlexBurst360 therapy, is yet another testament of that commitment.”
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
